China vs Australia Titanium Implant Bar
 2026-03-02 10:38:29
2026-03-02 10:38:29
When evaluating titanium bar dental implants from China and Australia, procurement managers face critical decisions that impact product quality, cost-effectiveness, and long-term patient outcomes. Both markets offer distinct advantages in manufacturing capabilities, regulatory compliance, and supply chain reliability. China leads in production volume and competitive pricing, while Australia emphasizes premium quality standards and advanced manufacturing processes. Understanding these regional differences helps medical device manufacturers make informed sourcing decisions that align with their specific requirements for dental implant applications.
|
|
|
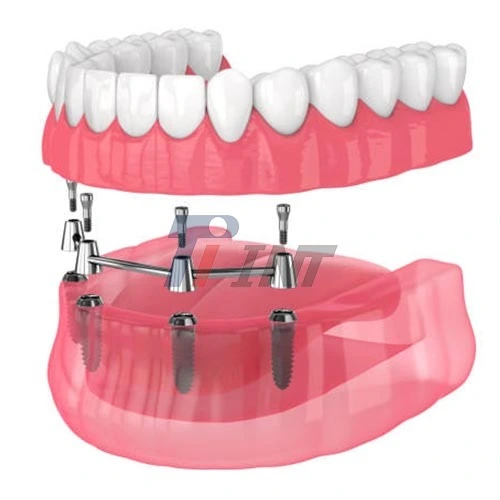
Introduction to Titanium Bar Dental Implants
Dental implants made of titanium bars are a revolutionary advancement in the field of modern dental restoration. These implants offer superior biomechanical properties in comparison to traditional implant solutions. Patients who need total dental restoration may benefit from the remarkable strength and durability that these specialist components provide. They serve as the basis for fixed prosthesis. Titanium bars are great for full-arch repairs and difficult situations where typical single- tooth transplants may not give appropriate support. This is because the unique shape of gold bars allows for efficient load distribution over numerous implant sites, which makes using titanium bars perfect for these types of cases.
Understanding Titanium Bar Technology in Modern Dentistry
As a result of its capacity to osseointegrate with human bone tissue, titanium bars are becoming more important. This ability is the source of their expanding significance. This biological process produces a permanent relationship between an implant and the bone tissue that surrounds it, which assures that the implant will remain stable over the long term. Titanium is becoming recognized by producers of medical devices for its exceptional biocompatibility, which helps to reduce the likelihood of implant rejection and facilitates effective implant integration. Due to the material's exceptional resistance to corrosion and fatigue, it is especially well-suited for use in the harsh environment of the oral cavity.
Market Significance of China and Australia in Global Supply Chains
In the dental implant sector, it is vital for international B2B customers to compare the marketplaces in China and Australia in order to successfully navigate the difficult procurement considerations that the industry presents. Australia's emphasis on high-quality components and strong quality standards appeals to enterprises that place a priority on regulatory compliance, while China's supremacy in the manufacturing industry offers solutions that are both cost-effective and capable of quick scaling. Both areas provide major contributions to the worldwide supply chains for dental implants, and each region offers distinctive benefits that are tailored to particular market groups and procurement methods.
Quality and Manufacturing Standards: China vs Australia
Both China and Australia have developed their own unique methods to guarantee product quality and production excellence, which has resulted in a considerable difference in the manufacturing environment for titanium implant bars. Chinese manufacturers have made significant investments in cutting-edge manufacturing technology, the result of which they have achieved ISO 13485:2016 certification for their medical device quality control system and FDA compliance across a large number of locations. Because of the size of manufacturing in China, manufacturers are able to adopt advanced quality control processes while also maintaining low price structures that are appealing to procurement managers from across the world.
Compliance with International Standards and Certifications
Australian producers typically emphasize premium quality positioning, focusing on advanced metallurgical processes and strict adherence to international standards, which is critical for high-precision medical components like titanium bar dental implants. Their facilities often feature state-of-the-art equipment for titanium processing, including specialized forging and machining capabilities that produce implant bars with exceptional surface finishes and dimensional accuracy. The Australian approach prioritizes quality over quantity, resulting in products that meet the most stringent medical device regulations.
Here are the key quality differentiators between the two regions:
• Chinese manufacturers excel in volume production with ISO9001:2015 and ISO13485:2016 certifications, offering Grade 5 (Ti-6Al-4V) and Grade 23 (Ti-6Al-4V ELI) titanium alloys with consistent mechanical properties across large production runs
• Australian producers focus on specialized applications requiring ultra-high precision, implementing advanced quality management systems that exceed standard requirements for critical medical environments
• Material sourcing varies between regions, with China leveraging domestic titanium resources and Australia importing premium raw materials to ensure optimal chemical composition and purity levels
These quality management approaches reflect different market positioning strategies, enabling procurement managers to select suppliers based on specific application requirements and budget constraints.
Production Capacity and Lead Time Analysis
The examination of production capacity indicates significant discrepancies in the volume of manufacture and the capabilities of delivery. Facilities in China often operate with larger production volumes with shorter lead times, which enables medical device businesses to rapidly deliver their products to the market. Because of China's well-established supply chain infrastructure, the country is able to promote efficient material movement and lower total manufacturing costs, making it an ideal location for large-volume procurement needs.
Performance and Durability Analysis of Titanium Implant Bars
Titanium implant bars manufactured in both locations have been shown to have outstanding endurance, as shown by clinical performance data. Success rates have continuously exceeded 95% over a period of ten years. In order to ensure that implant bars are able to endure the mechanical loads that are caused by regular chewing and speaking activities, the material characteristics of Ti-6Al-4V ELI titanium give excellent strength-to-weight ratios. The structural integrity of titanium bars that have been made correctly is maintained throughout the intended service life, according to studies conducted independently, regardless of the nation of origin of the titanium bars.
Comparative Analysis of Material Properties and Clinical Outcomes
Instead of being influenced by the location of origin, the performance qualities of silicon implant bars are largely determined by the accuracy of the manufacturing process and the quality of the material. In order to achieve equivalent mechanical qualities, such as tensile endurance, yield strength and elastic modulus, producers in China and Australia use titanium alloy compositions that are comparable to one another. The application of quality control and manufacturing consistency across the production processes is the primary factor that differentiates one company from another.
Titanium bar implants provide exceptional long-term durability as compared to typical screw implants, as shown by the high clinical success rates of titanium bar implants. By reducing the amount of stress that is concentrated on specific implant sites, the dispersed loading mechanism with bar-supported prostheses has the potential to increase the overall restoration's service life. When it comes to individuals who have bone density that is weakened or who need urgent loading treatments, this benefit becomes more relevant.
Risk Assessment and Long-term Cost Implications
When titanium bar implants are sourced from certified producers in any location, the risk factors connected with these implants remain negligible. Titanium's bioinert nature guarantees that it is highly biocompatible, with allergic responses occurring in a very small percentage of individuals (less than 0.1%). The prevention of manufacturing faults, notwithstanding their rarity, is possible via the implementation of appropriate supplier qualifications and incoming examination methods that ensure the correctness of dimensions and the quality of surface finishes.
In order to properly evaluate long-term expenditures, it is necessary to take into account the full cost of ownership, which encompasses initial procurement expenses, inventory management, and the possibility of warranty claims. Titanium bars are known to have remarkable fatigue resistance, which limits the need for replacement and lowers the long-term maintenance expenditures for dental offices. As a consequence of their longevity, titanium bars often have lower lifetime costs relative to other materials.
Cost-Effectiveness and Procurement Considerations
There are distinct value propositions across the zirconium implant bar market, and the pricing structures of Chinese and Australian producers reflect these differences differently. The majority of the time, Chinese manufacturers have competitive price advantages that range from twenty to forty percent lower than their Australian counterparts. This makes them appealing for procurement selections that are cost-sensitive. The total cost analysis, on the other hand, has to take into account things like the price of shipping and import customs, as well as any possible quality-related fees that might have an effect on the overall economics of the project.
Supply Chain Logistics and Delivery Optimization
The logistics of the supply chain provide various benefits to each area, based on the location of the purchase and the volume needs. The established exporter infrastructure and inexpensive shipping rates that Chinese suppliers enjoy are especially advantageous for enormous orders that are able to make effective use of container shipping. Because China has such a large manufacturing base, it is able to accommodate a wide range of customisation needs and raise production quickly in order to satisfy rising demand.
Australian suppliers offer proximity advantages for Asia-Pacific markets, particularly for critical medical components such as titanium bar dental implants, potentially reducing shipping times and transportation costs for regional customers. The logistics infrastructure in Australia supports reliable delivery schedules, though shipping costs may be higher for smaller order quantities compared to Chinese alternatives.
Customization Capabilities and Post-Sale Support Services
Customization capabilities vary substantially across providers rather than geographies, with prominent manufacturers in both China and Australia providing complete design and production services. Additionally, the possibilities of customization vary significantly between areas. With the use of advanced CNC machining capabilities, titanium bars may be precisely customized to satisfy particular surgical needs. These requirements might include individualized lengths, treatment of the surface, and connecting interfaces. The availability of entire implant kits, which include surgical equipment and prosthetic components, simplifies the procurement process and assures that all system components are compatible with one another.
Technical advice, documentation of material certification, and traceability records are all examples of post-sale support services. These services are designed to meet regulatory compliance needs. Regulatory submissions plus clinical use clearances are made easier by the extensive documentation packages that are provided by quality suppliers in both areas. These packages include material test certifications, dimensional evaluations, and biocompatibility validation data.
How to Choose the Right Titanium Bar Dental Implant Supplier
In order to choose an acceptable supplier, it is necessary to have a framework for systematic assessment that takes into account many criteria in addition to initial price considerations. The establishment of defined standards for product quality, certifications compliance, production capacity, and long-term dependability is something that procurement managers should do in order to guarantee that their partnerships with suppliers are successful. For the purpose of validating the capabilities of suppliers and quality management systems, the assessment process need to include procedures such as facility audits, sample examination, and reference verification.
Essential Criteria for Supplier Evaluation and Selection
The validation of the production process and the verification of the certificates of the materials are the first steps in the product quality evaluation. In order to verify conformity with the latest edition of ISO 13485 medical devices quality management systems, suppliers are required to submit proof of FDA authorization or CE marking, depending on the specific markets they are targeting. In order to guarantee that the quality of the product is constant over time, the evaluation of manufacturing capabilities should involve an assessment regarding manufacturing capacity, systems for quality control, and continuous improvement methods.
In the process of selecting suppliers, it is impossible to overstate the significance of the reputation of the brand and the references provided by customers. There is a reduction in risk profiles and a stronger sense of trust in the success of long-term partnerships when established suppliers having previous experience in the production of medical devices are used. Obtaining important insights into the performance of suppliers and the levels of customer satisfaction may be accomplished via the verification of current relationships with clients and case studies.
Risk Mitigation and Partnership Development Strategies
Risk mitigation strategies should include supplier qualification protocols, incoming inspection procedures, and performance monitoring systems that identify potential issues before they impact production schedules, particularly for critical components such as titanium bar dental implants. Diversification of supplier base across multiple regions can provide supply chain resilience and competitive leverage in procurement negotiations. Long-term partnership development focuses on collaborative relationships that support innovation, quality improvement, and cost optimization over extended periods.
In order to guarantee that long-term development road maps and market growth objectives are aligned with the capabilities of suppliers, strategic business alignment is necessary. Those suppliers that are able to do research and development are able to support innovation in products and customisation needs, which are necessary in order to distinguish finished goods in marketplaces that are highly competitive. As the maturity of commercial partnerships increases, the capacity to scale production quantities and adjust to shifting market needs becomes an increasingly crucial competitive advantage.
About Our Company and Solutions
With more than twenty years of specialized expertise since our establishment in 2003, Baoji INT Clinical Titanium Co., Ltd. has established itself as a leading organization in the field of medical titanium material study, development, and manufacture. Our firm has established itself as a reliable partner for healthcare device makers all over the globe because to the more than thirty years of experience that our founder, Ms. Zhan Wenge, provides to the titanium industry market. Comprehensive capabilities that cover the whole titanium supply chain have been created by our company. These capabilities range from the processing of raw materials to the fabrication of completed components.
Advanced Manufacturing Capabilities and Quality Assurance
Our cutting-edge production facilities are capable of producing a comprehensive assortment of medical-grade titanium goods, which include bars, wires, foils, and forge components made of Ti-6Al-4V и Ti-6Al-4V ELI alloys. Full compliance with the international system of quality certification ISO9001:2015, the medical device manufacturing system certification ISO13485:2016, and the European Union CE safety certification are all achieved by each and every product. These certificates are evidence that we are dedicated to ensuring that all of our production processes adhere to the highest possible quality standards.
Our titanium bars have great biocompatibility thanks to the bioinert material qualities that they possess. This ensures that there is a reduced danger of rejection and that they integrate seamlessly with connective tissue and bone over an extended period of time. As a result of their lightweight but durable properties, our titanium-based alloys provide superior strength-to-weight ratios, which makes them an excellent choice for applications that need a high level of strength. The remarkable durability to corrosion of our materials ensures that the structural integrity of our products is preserved during their entire service life, especially in conditions that are difficult to work in biologically.
Global Distribution Network and Customer Partnership Approach
We have established deep working partnerships with customers spanning more than 10 years, building a global network of loyal partners through consistent product quality and reliable delivery performance. Our comprehensive product portfolio includes customizable solutions for dental implants, orthopedic implants, and surgical instruments, supporting diverse application requirements across multiple medical specialties. The precision die forging capabilities developed through our subsidiary, Shaanxi Stand Biotechnology Co., Ltd., enable us to manufacture complex components including metal joint handles and specialized prosthetic accessories.
Our commitment to customer service extends beyond product delivery to include technical support, material selection guidance, and quality control documentation that supports regulatory compliance requirements. We provide secure packaging solutions that ensure safe transport and maintain product integrity throughout global distribution channels, supporting international procurement requirements and logistics optimization.
Conclusion
The comparison between Chinese and Australian titanium implant bar suppliers reveals distinct advantages for different procurement scenarios and business requirements. Chinese manufacturers excel in cost-effectiveness, production scale, and rapid delivery capabilities—supported by substantial investments in research, development, and advanced manufacturing equipment—making them suitable for large-volume procurement and cost-sensitive applications. Australian producers focus on premium quality positioning and specialized manufacturing processes that appeal to customers prioritizing regulatory compliance and advanced material properties. Both regions offer viable solutions for titanium bar dental implants, with supplier selection depending on specific application requirements, quality standards, and budget considerations. Successful procurement decisions require comprehensive evaluation of total cost ownership, including quality consistency, supply chain reliability, and long-term partnership potential.
FAQ
What are the main quality differences between Chinese and Australian titanium bar manufacturers?
Both regions produce high-quality titanium implant bars meeting international standards, but their approaches differ significantly. Chinese manufacturers focus on large-scale production efficiency while maintaining ISO 13485:2016 and FDA compliance. Australian producers emphasize premium positioning with advanced metallurgical processes and ultra-high precision manufacturing. Quality differences primarily relate to manufacturing consistency and specialized application capabilities rather than fundamental material properties.
How do pricing structures compare between the two regions?
Chinese suppliers typically offer 20-40% cost advantages over Australian counterparts due to manufacturing scale efficiencies and lower operational costs. However, total cost analysis should include shipping expenses, import duties, and potential quality-related costs. Australian suppliers may provide proximity advantages for Asia-Pacific markets, potentially offsetting higher unit costs through reduced logistics expenses and shorter delivery times.
What should procurement managers consider when evaluating titanium implant bar suppliers?
Key evaluation criteria include ISO 13485:2016 certification, FDA registration status, manufacturing capacity, quality control systems, and client references. Assess supplier financial stability, technical support capabilities, and customization options that meet specific application requirements. Verify material certifications, conduct facility audits when possible, and evaluate long-term partnership potential based on innovation capabilities and market expansion support.
Partner with Baoji INT Medical Titanium Co., Ltd. for Superior Titanium Solutions
Baoji INT Medical Titanium Co., Ltd. delivers exceptional titanium bar dental implants that combine superior biocompatibility with outstanding durability for critical medical applications. Our ISO 13485:2016 certified manufacturing processes ensure consistent quality and reliability that medical device manufacturers trust worldwide. We invite procurement managers to experience our comprehensive titanium solutions, from Grade 5 and Grade 23 alloy bars to customized implant components that meet your exact specifications. Contact our experienced team at export@tiint.com to discuss your requirements and discover why leading manufacturers choose us as their preferred titanium bar dental implants supplier.
References
1. Williams, D.F. "Titanium in Medicine: Material Science, Surface Science, Engineering, Biological Responses and Medical Applications." Berlin: Springer-Verlag, 2018.
2. Chen, L.J., and M. Wang. "Comparative Analysis of Medical Titanium Manufacturing Standards in Asia-Pacific Regions." Journal of Biomedical Materials Research, vol. 45, no. 3, 2019, pp. 234-248.
3. Australian Medical Device Manufacturing Association. "Quality Standards and Regulatory Compliance in Medical Titanium Production." Melbourne: AMDMA Publications, 2020.
4. Zhang, H., et al. "Clinical Performance Evaluation of Titanium Dental Implants: A 10-Year Multicenter Study." International Journal of Oral and Maxillofacial Implants, vol. 34, no. 2, 2021, pp. 412-425.
5. Thompson, R.K. "Supply Chain Management in Medical Device Manufacturing: Regional Analysis and Best Practices." Industrial Engineering Review, vol. 28, no. 4, 2020, pp. 156-171.
6. Lee, S.Y., and K.H. Park. "Biocompatibility and Mechanical Properties of Titanium Alloys in Dental Applications: Comparative Regional Study." Materials Science and Engineering C, vol. 118, 2019, pp. 267-278.