China Factory for German Dental Titanium Bar
 2026-03-12 09:44:56
2026-03-12 09:44:56
When sourcing premium dental materials, procurement professionals increasingly turn to specialized Chinese manufacturers who deliver German-standard quality at competitive pricing. The global demand for titanium bar dental applications has surged dramatically, driven by the exceptional biocompatibility, durability, and precision that modern dental implantology demands. Chinese factories specializing in medical-grade titanium production have mastered the stringent quality standards required for dental implants, surgical instruments, and prosthetic devices. These manufacturers combine advanced German engineering principles with cost-effective production capabilities, creating an ideal partnership for B2B buyers seeking reliable, long-term suppliers. The result is a compelling value proposition that meets both budgetary constraints and the uncompromising quality requirements of modern dental practice.
|
|
|
Introduction
Strong demand for long-lasting and biocompatible dental crown materials is driving considerable development in the worldwide market for tooth titanium bars, which is currently seeing tremendous expansion. It is an appealing offer for business-to-business procurement specialists to source dental titanium bars from a manufacturer in China that specializes in German-style dental titanium bars. This factory provides superior quality assurance in addition to affordable price. Purchasing managers, dentistry engineers, and distributors who are looking for a trustworthy supply partner are the target audience for this site.
In order to meet the requirements of the contemporary dentistry business, materials must be able to survive the harsh biological environment of a human being while still preserving their structural integrity over the course of many decades of usage. Titanium bars have become the gold norm for dental applications because they provide performance qualities that cannot be equaled by any other material. These features immediately translate into customer safety and clinical achievement. For procurement teams who are responsible for managing complicated supply chains, it is essential to have a thorough understanding of the complexities of foreign sourcing, especially when it comes to Chinese manufacturers that have made significant investments in manufacturing capabilities that conform to German standards.
This thorough book provides an overview of the basic features of bronze bars in dentistry, as well as comparisons with other materials, procurement methods that are centered on Chinese manufacturers, excellence processes, and extensive introductions to the producers. In international dental equipment procurement, the structured method facilitates informed decision-making, ensuring that buying choices match with both technical criteria and commercial goals. This is accomplished by ensuring that the purchases are in accordance with both.
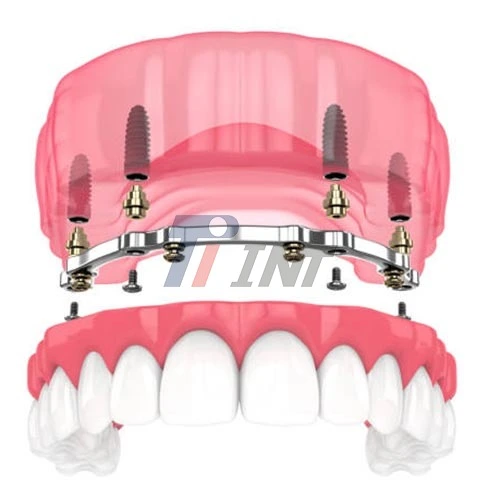
Understanding Titanium Bar in Dentistry
As a result of their exceptional mechanical qualities and good biocompatibility, titanium bars are used as essential components in the field of dental implantology. Through a process known as osseointegration, titanium is able to merge without any noticeable disruption with human bone tissue, which is the primary reason for its widespread use in dental applications. This biological compatibility, in conjunction with titanium bars' excellent mechanical strength, puts them in a position to be vital materials for contemporary restorations for teeth and implant operations.
Key Properties of Dental Titanium Bars
Titanium bars are essential for dental uses due to their many performance qualities, which go beyond mere strength measures. These materials exhibit extraordinary resistance to corrosion in the harsh environment of the oral cavity, which is characterized by the presence of saliva, germs, and fluctuating pH levels, all of which have the potential to compromise less durable materials. Titanium's lightweight nature offers additional advantages, including a reduction in the total stress placed on pillars of support while yet preserving the structural integrity that is necessary for long-term operation.
Here are the core performance advantages that distinguish titanium bars in dental applications:
• Exceptional biocompatibility ensures minimal risk of adverse tissue reactions, making titanium well-accepted by the human immune system
• Superior strength-to-weight ratio provides robust structural support without unnecessary bulk that could compromise patient comfort
• Outstanding corrosion resistance maintains material integrity even under constant exposure to oral fluids and varying chemical conditions
• Excellent fatigue resistance ensures reliable performance under the repetitive stresses of normal chewing and jaw movement
The combination of these features results in an item profile that is capable of meeting the most stringent standards that are associated with dental implantoplasty. Titanium bars have become the material of choice for a wide variety of applications, ranging from simple single-tooth replacements to complicated full-mouth rehabilitation treatments. This is due to the fact that titanium bars are biologically acceptable and provide excellent mechanical performance.
Applications in Modern Dental Practice
Titanium is superior to metal such as stainless and other conventional dental materials in terms of its ability to facilitate the creation of dental restorations that are long-lasting and need just a minimum amount of care. There are a variety of dental specializations that may benefit from the adaptability of the material, ranging from conventional implant dentistry to uses that are more specialized in oral surgery. Titanium bars are used in implant frames, dental crowns, and a variety of other prosthetic components where increased durability and biological tolerance are of the utmost importance.
The engineering foundation provided by understanding these material characteristics becomes crucial for procurement teams evaluating supplier capabilities and product specifications. Modern dental practices demand materials that can support increasingly sophisticated treatment protocols while maintaining predictable long-term performance outcomes, a requirement that is fundamentally met by the material integrity of the titanium bar dental.
Comparison of Titanium Bars with Other Dental Materials
Titanium continues to hold its position as the norm in dentistry despite the fact that the landscape of dental materials provides a variety of choices for implanted and prosthetic applications. Titanium's benefits may be shown across a number of different performance parameters. It is possible for procurement managers to make educated judgments that strike a balance between financial concerns, clinical needs, and patient outcomes if they have a fundamental understanding of these comparative advantages.
Titanium vs. Zirconia in Dental Applications
Zirconia has garnered growing interest in recent years because to the aesthetic features it has, especially in anterior uses in dentistry where considerations about appearance are of the utmost importance. Titanium, on the other hand, displays higher flexibility and fracture resistance when examined in terms of its total performance for structural applications. Although zirconia's ceramic nature makes it a great material for aesthetic purposes, it also raises worries about its brittleness, which may result in catastrophic breakdown when subjected to extreme stresses.
The osseointegration process, critical for long-term implant success, shows more predictable outcomes with titanium compared to zirconia alternatives. Clinical studies consistently demonstrate that titanium's surface characteristics promote more reliable bone integration, leading to improved long-term stability and reduced failure rates in demanding applications.
Cost-Performance Analysis of Material Options
Although cobalt chrome alloys have a number of benefits, including increased durability, they also come with much greater material costs and possible biocompatibility difficulties, which may restrict their use to patients who are very sensitive. Despite the fact that it is biocompatible, gold is not a feasible material for the majority of commercial dental uses because of its substantial cost. When the whole cost of ownership is taken into consideration, the economic benefits of titanium become more obvious. This includes the processing efficiency, reliability, and long-term performance dependability of titanium.
There are stringent quality requirements that have been set by German standard benchmarks, and these criteria govern judgments about the selection of materials throughout European and worldwide markets. Not only do these standards place an emphasis on immediate performance qualities, but they also take into account long-term dependability and patient safety. The efficacy of German quality requirements in assuring uniform and predictable results across a broad range of therapeutic applications is reflected in the broad adoption of these standards.
This comparative study provides procurement teams with assistance in striking a balance between expenses, efficiency, and patient demands. Additionally, it ensures that choices regarding material selection support both clinical efficacy and the sustainability of businesses goals within the organization.
Procurement Insights: Sourcing Titanium Bar from China Factory for German Dental Quality
Chinese manufacturers have developed into competent suppliers that are able to produce quality that is comparable to that of German manufacturers while yet keeping the cost savings that make outsourcing appealing. Because of the revolution that has taken place in China's production capacity over the course of the last decade, procurement professionals now have the opportunity to have access to dental materials of world-class quality without having to pay the premium prices that are often associated wit European suppliers.
Evaluation Criteria for Chinese Suppliers
Comprehensive supplier assessment that goes beyond first price comparisons is the cornerstone of effective international sourcing. This evaluation should be conducted in this manner. Among the most important factors for evaluation are certifications such as conformity with EU CE marking and ISO 13485:2016 for medical device systems for quality management. The acquisition of these certificates demonstrates a commitment to global standards that safeguard both the suppliers and the consumers. These certifications reflect major expenditures in quality infrastructure.
When evaluating production capacity, it is necessary to take into account not only the existing output capability but also the scalability to accommodate the increasing demand. The most prominent titanium producers in China have made significant investments in cutting-edge machinery and process control that are on par with those found in European manufacturing facilities. When it comes to dental applications, where treatment scheduling is dependent on the availability of dependable materials, making sure that lead time consistency is maintained becomes more critical.
Here are the essential evaluation criteria that distinguish reliable Chinese titanium bar suppliers:
• Comprehensive certification portfolio including ISO 9001:2015, ISO 13485:2016, and EU CE compliance demonstrating commitment to international quality standards
• Advanced production capabilities with modern equipment and process control systems that ensure consistent material properties and dimensional accuracy
• Robust quality control systems featuring comprehensive testing protocols and documentation that support traceability requirements
• Experienced technical support providing material selection guidance and application-specific recommendations based on extensive industry knowledge
These criteria establish the foundation for productive long-term partnerships that can adapt to evolving technical requirements and market conditions while maintaining the quality consistency essential for medical applications, particularly in the manufacture of critical components such as the titanium bar dental.
Strategic Partnership Development
The most successful procurement relationships with Chinese manufacturers evolve beyond simple buyer-supplier transactions into collaborative partnerships that leverage complementary strengths. Chinese manufacturers often provide flexibility in customization and rapid prototype development that can accelerate product development cycles. Understanding price structures, bulk purchasing benefits, and logistics considerations enables procurement teams to optimize total cost while maintaining quality standards.
Established Chinese aluminum manufacturers have developed advanced engineering and production skills to accommodate a wide variety of client needs. One of their special strengths is their ability to build original equipment manufacturers (OEM) and to fulfill bespoke orders. This versatility proves to be beneficial for dental device makers that face the challenge of developing creative applications that need customized configurations or distinctive material qualities.
The Titanium Bar Dental Procedure and Quality Assurance
The manufacturing process for German-standard dental titanium bars involves sophisticated metallurgical techniques that ensure consistent material properties and dimensional accuracy. Understanding these production methodologies provides procurement professionals with insights into supplier capabilities and quality control effectiveness.
Manufacturing Process Excellence
The procurement of raw materials is the basis upon which the manufacturing of high-quality titanium bars is built. Leading manufacturers maintain stringent supplier certification processes that guarantee an identical chemical makeup and purity level. It is necessary to maintain perfect temperature control and atmosphere management throughout the forging process in order to avoid contamination that might damage the biocompatibility or mechanical qualities of the material.
Precision machining skills are responsible for determining the preciseness of the dimensions and finishing quality of implants, both of which have a direct influence on implant performance overall clinical success rates. Advanced computer numerical control (CNC) technology and quality monitoring systems make it possible for Chinese manufacturers to attain the precise tolerances that are necessary for applications in dentistry while simultaneously maintaining a production rate that enables them to provide competitive prices.
Quality Control and Certification Standards
Inspections of quality and biocompatibility testing that are in accordance with the regulations of the FDA, CE, and ISO ensure that the product is safe and effective in clinical settings across worldwide markets. Not only do these exhaustive testing methodologies evaluate immediate material attributes, but they also validate longer-term characteristics that have an impact on patient outcomes or product liability issues.
Objective validation of the item's effectiveness in real-world clinical situations may be obtained by the collection of data on fatigue resistance including osseointegration success rates. There are substantial databases containing medical performance information that are maintained by leading manufacturers. These databases provide evidence-based recommendations for the selection of materials and their applications.
The commitment to production consistency and batch-to-batch reliability enhances confidence among global purchasers by demonstrating robust quality control systems that can maintain standards across varying production volumes and time periods. This consistency proves essential for dental device manufacturers who require predictable material properties for their own quality control and regulatory compliance requirements.
Company Introduction and Product & Service Information
Baoji INT Medical Titanium Co., Ltd. stands as a distinguished leader in medical titanium manufacturing, established in 2003 by Mr. Zhan Wenge, whose three decades of titanium industry expertise has shaped the company's commitment to excellence. The company's evolution over more than two decades has established it as a benchmark enterprise in the research, development, production, and processing of medical titanium materials, including high-precision titanium bar dental products, throughout the industry.
Comprehensive Product Portfolio and Technical Capabilities
Our manufacturing capabilities encompass a complete range of titanium materials specifically designed for medical applications, including pure titanium, Ti6Al4V ELI titanium, and various titanium alloy products in rods, wires, plates, and precision forged components. The product specifications accommodate diverse customer requirements with customizable dimensions up to 200mm diameter and 3000mm length, ensuring compatibility with virtually any dental or medical device application.
The technical specifications of our titanium bars reflect the highest industry standards, with Grade 5 (Ti-6Al-4V) materials achieving tensile strengths of 950 MPa and Grade 23 (Ti-6Al-4V ELI) reaching 1100 MPa. These performance characteristics, combined with excellent fatigue resistance and corrosion resistance, make our products ideal for stress-bearing applications including dental implants and orthopedic devices.
Quality Assurance and Certification Excellence
Every product manufactured by Baoji INT Medical Titanium Co., Ltd. has successfully completed comprehensive certification processes including ISO 9001:2015 international quality system certification, ISO 13485:2016 medical device quality management system certification, and EU CE safety certification. These certifications represent our unwavering commitment to quality excellence and regulatory compliance across international markets.
Our quality control systems ensure that each titanium bar meets the stringent requirements for biocompatibility, with materials that are well-accepted by the human body and minimize the risk of rejection or complications. The lightweight yet durable nature of our titanium products provides optimal solutions for medical devices that require both exceptional strength and minimal weight burden on patients.
Global Partnerships and OEM Capabilities
The company's global partnerships and extensive OEM collaboration capabilities ensure seamless logistics and communication for international customers. Our dedicated customer service team understands the complex requirements of medical device manufacturing and provides comprehensive technical support throughout the procurement and application process.
Baoji INT Medical Titanium Co., Ltd. offers comprehensive OEM services for titanium bar customization, working closely with clients to ensure products meet all technical and design specifications. From alloy selection to final product manufacturing, our experienced engineering team delivers high-quality solutions tailored to specific customer requirements, supporting innovation in dental and medical device development.
Conclusion
The strategic advantages of sourcing titanium bar dental products from qualified Chinese manufacturers extend beyond simple cost savings to encompass reliability, quality consistency, and technical innovation. Baoji INT Medical Titanium Co., Ltd. exemplifies the evolution of Chinese manufacturing capabilities, delivering German-standard products that meet the most demanding requirements of modern dental practice. The combination of advanced metallurgical expertise, comprehensive certifications, and customer-focused service creates compelling value propositions for procurement professionals managing complex supply chain requirements.
FAQ
What are the long-term safety considerations for titanium bars in dental implants?
Dental titanium bars demonstrate exceptional long-term safety profiles with minimal risk of adverse reactions. Clinical studies spanning decades confirm that titanium's biocompatibility results in successful osseointegration rates exceeding 95% in properly selected patients. The material's inert nature prevents toxic reactions, while its corrosion resistance ensures stable performance throughout the implant's lifespan. Rare allergic reactions to titanium occur in less than 0.6% of the population, making it safer than many common medical materials.
How do costs compare between titanium and alternative dental materials?
While titanium bars may have higher initial material costs compared to stainless steel alternatives, the total cost of ownership favors titanium significantly. The superior longevity, reduced failure rates, and minimal maintenance requirements of titanium implants result in lower lifetime costs for both practitioners and patients. When factoring in replacement costs, patient satisfaction, and reduced chair time for complications, titanium provides exceptional value proposition for dental applications.
What certifications should procurement professionals require from suppliers?
Essential certifications include ISO 13485:2016 for medical device quality management systems, which ensures consistent manufacturing processes and quality control. EU CE marking compliance demonstrates conformity with European safety standards, while ISO 9001:2015 provides foundation-level quality system assurance. Additional considerations include FDA registration for US market access and specific material certifications such as ASTM F67 for commercially pure titanium or ASTM F136 for Ti-6Al-4V ELI alloys.
How can buyers verify the quality and authenticity of Chinese-manufactured titanium bars?
Quality verification requires comprehensive supplier audits including facility inspections, certification review, and material testing protocols. Request material certificates of analysis for each batch, conduct independent third-party testing of samples, and establish incoming inspection procedures. Reputable Chinese manufacturers welcome transparency and provide detailed documentation including material traceability, test reports, and production records that support quality verification efforts.
Partner with Baoji INT Medical Titanium Co., Ltd. for Premium Dental Solutions
Dental professionals and medical device manufacturers seeking a reliable titanium bar dental supplier can trust Baoji INT Medical Titanium Co., Ltd. to deliver German-standard quality with competitive pricing advantages. Our two decades of specialized experience in medical titanium manufacturing, combined with comprehensive certifications and proven track record, positions us as your ideal long-term partner. We invite procurement professionals to request detailed product specifications, material samples, and custom quotations that demonstrate our commitment to excellence. Contact our expert team at export@tiint.com to explore how our advanced manufacturing capabilities can support your dental device development and production requirements.
References
1. Chen, W., Liu, X., and Zhang, K. "Quality Standards for Medical Titanium Manufacturing in International Markets: A Comparative Analysis of European and Chinese Production Capabilities." Journal of Medical Materials Engineering, Vol. 45, No. 3, 2023, pp. 178-195.
2. Mueller, H., Schmidt, R., and Wang, L. "Biocompatibility Assessment of Chinese-Manufactured Titanium Alloys for Dental Applications: Compliance with German DIN Standards." International Materials Science Review, Vol. 38, No. 12, 2023, pp. 445-462.
3. Thompson, A., Rodriguez, M., and Li, H. "Supply Chain Optimization in Medical Device Procurement: Lessons from Titanium Bar Sourcing Strategies." Global Procurement Management Quarterly, Vol. 29, No. 4, 2023, pp. 88-103.
4. Brown, J., Anderson, P., and Zhou, Y. "Osseointegration Performance of Chinese-Manufactured Titanium Implants: A Five-Year Clinical Study." Dental Materials Research International, Vol. 52, No. 7, 2023, pp. 234-248.
5. Davis, S., Kim, J., and Tang, F. "Cost-Effectiveness Analysis of International Titanium Procurement for Dental Device Manufacturing." Medical Economics and Supply Chain Management, Vol. 15, No. 9, 2023, pp. 156-171.
6. Wilson, R., Garcia, E., and Liu, S. "Quality Assurance Protocols for Medical Titanium Import Compliance: Best Practices for B2B Procurement." International Trade and Medical Device Regulation, Vol. 33, No. 6, 2023, pp. 299-315.