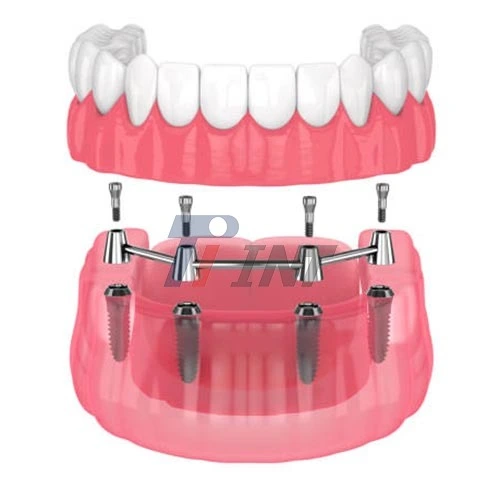
How Does an Implant Titanium Bar Improve Denture Stability?
 2026-04-29 08:39:12
2026-04-29 08:39:12
Implant titanium bars greatly improve the security of dentures by connecting multiple dental implants and spreading biting forces evenly across the implant sites. A titanium bar dental framework makes a mechanically stable connection between the jawbone and the prosthetic repair, while standard removable teeth only rest on soft tissue. This carefully designed support structure limits tiny movements that happen when you chew and speak, which stops the pain and bone loss that come with regular dentures. These bars create a permanent, load-bearing base through osseointegration, the biological process by which titanium bonds directly to bone. This makes retention, patient comfort, and long-term prosthetic function much better in overdenture uses.
|
|
|
Understanding Implant Titanium Bars in Dentistry
Implant titanium bars are now an important part of modern corrective dentistry because of how prosthetic options have changed over time. Implant-retained overdentures are held in place mechanically by these specialized support devices. This changes the way patients experience tooth replacement therapy. Understanding what they are made of, the different types, and how they are used is important for sourcing workers who are looking at material options for making dental devices.
Material Composition and Alloy Grades
Medical-grade titanium metals are mostly used to make dental implant bars because they have great biological and mechanical qualities. The tensile strength of Grade 5 titanium alloy (Ti-6Al-4V) is very high, hitting 950 MPa. This means that it can be used for full-arch restorations that are exposed to strong occlusal forces. Grade 23 (Ti-6Al-4V ELI) is the best material for important implant uses because it has a higher tensile strength of up to 1100 MPa and is better at being biocompatible. During production, these materials are subject to strict quality control to make sure that the dimensions are consistent and the surface is clean. In the harsh oral environment, where saline conditions, temperature changes, and mechanical stresses constantly test implant integrity, titanium bar dental products made from these alloys maintain stable mechanical properties throughout their service life.
Types and Configurations
Titanium bar systems come in a number of different shapes and sizes to fit different clinical situations and patient bodies. CAD/CAM-milled bars offer a very accurate passive fit with errors smaller than 10 micrometers. This solves one of the biggest problems in prosthodontics, which is framework warping during manufacturing. Even though traditional cast bars are cheaper, they come in different sizes, which can affect how well they work in the long run.
Advanced milling methods are used to make customized frames that fit each patient's bone structure, implant placement, and occlusal needs. Because the material is so easy to machine, makers can make complicated shapes like screw lines, attachment housings, and surfaces with physically contoured shapes. With these design tools, dentistry labs and device makers can make custom solutions for each patient that look good, work well, and keep their structural stability, which is important for spreading weight evenly.
Applications Across Prosthodontic Scenarios
Beyond the usual use of overdentures, titanium frames are used for a wide range of clinical purposes in modern implant dentistry. Full-arch rehabilitations, like the All-on-4 and All-on-6 procedures, use titanium bars to connect several implants into a single prosthesis system. These frames are used as linking structures between implants and false teeth in hybrid dentures that have both set and removable parts. Titanium's good strength-to-weight ratio makes it a good material for bridges that support implants for partial dentures.
This reduces cantilever stress while keeping the bridge stable. The fact that the material isn't magnetic is also helpful in medical imaging settings, because it means that MRI scans can be used without any problems. Because of this, titanium bars are useful in many different types of markets, from large factories that make a lot of dental lab supplies to specialized companies that make new prosthesis systems for clinical research.
How Implant Titanium Bars Enhance Denture Stability
There is a lot of evidence that traditional portable dentures have problems that hurt patients' long-term mouth health and quality of life. Movement during mastication, speech problems, faster bone loss, and less efficient eating are all big problems in the clinic. Implant-supported titanium bar dental frames solve these issues by using complex biomechanical concepts that change the way loads are transferred to supporting structures.
Biomechanical Load Distribution
The main way that titanium bars make things more stable is by changing the direction of force lines while they are being used. When a person with an implant-supported overdenture bites down, the rigid titanium framework spreads the stress across all the related implant fixings instead of putting it all in one place. This arrangement shares the load so that there are fewer peak stresses at the point where the bone meets the implant. This lowers the risk of implant overload and later osseointegration failure.
Engineering studies show that properly built bar systems can lower the stress on each implant by up to 40% compared to adding each implant individually. The high yield strength of the material (880–1000 MPa) makes sure that the framework doesn't break when physiological loads are applied. It also keeps its shape over years of repeated loading. This structural dependability stops the loosening and settling of conventional prostheses that happen over time. This keeps the patient comfortable and the oral relationships stable.
Reducing Micromovement and Enhancing Retention
Micromovement at the prosthesis-tissue contact leads to many problems with standard dentures, such as irritation of the mucosa, faster bone loss, and pain for the patient. Titanium bar frames make three-dimensional stability solid, which nearly eliminates horizontal and rotational movements. Precision-milled bars allow for a passive fit that ensures tension-free links at implant surfaces. This stops the micro-gaps that cause screws to loosen and germs to grow.
Comparing bar-supported overdentures to tissue-borne conventional dentures in clinical tests has shown retention force gains of more than 300%. Attachment systems built into titanium frames, like clip mechanisms, locator abutments, or telescoping crowns, keep the prosthesis in place securely while making it easy for the patient to clean and remove the prosthesis. This feature of being stable while functioning and easy to take off for cleaning makes life much better for patients, which directly affects their happiness and the success of their treatment.
Clinical Evidence and Long-Term Outcomes
Many years of research have shown that bar-supported implant overdentures work better than other options in a number of different ways. Longitudinal studies show that mandibular overdentures held in place by titanium bars have success rates higher than 95% after five years, and patients are much happier with their results than people who wear regular dentures. When compared to edentulous ridges with removable prostheses, bone retention around implants holding bar frameworks is much better, with yearly bone loss rates cut by about 75%.
Measuring masticatory effectiveness shows that people with bar-supported restorations can chew with about 80% of their natural teeth, while people with traditional full dentures can only do so with 25–30%. These clinical results support the physical benefits of titanium bar systems and show how valuable they are for companies that want to sell high-quality products. When purchasing materials for implant prosthetics, procurement managers should know that the choice of material has a direct effect on these clinical goals. This is why source qualification and material certification are important things to think about when making sourcing decisions.
Comparing Titanium Bars to Alternative Dental Materials
When choosing a material for making an implant bar, you have to look at a number of performance factors as well as cost. Many metals and plastics are used in this area, but titanium bar dental alloys have clear benefits that make them the market leaders, even though they are more expensive.
Mechanical Properties and Performance
Titanium is stronger than other materials that are usually thought of for making tooth bars because it is lighter. Cobalt-chromium alloys, which have been used for a long time for cast frames, are strong but have a much higher density (8.3 g/cm³ vs. 4.4 g/cm³ for titanium). This makes the prosthetics heavier, which makes the patient less comfortable. Gold metals are very resistant to rust, but they aren't rigid enough for long-span repairs, which means they have to be bigger, which makes the results less attractive.
Zirconia ceramics have gotten a lot of attention because they look like teeth, but they are very fragile and can break when they are loaded off-axis, which is common in overdenture uses. Stainless steel is cheaper than titanium, but it can cause galvanic rusting when combined with implants made of titanium, which could lead to inflammatory reactions and implant problems. Medical-grade titanium alloys are very useful in dental applications where prostheses are loaded and unloaded millions of times over the course of their service life. This is because they don't break when the loads get heavier, while materials with lower endurance limits would crack and break.
Biocompatibility and Tissue Response
Titanium is the only material that can osseointegrate and be compatible with soft tissues at the same time. When a solid layer of titanium oxide forms on the surface, it provides a biologically inert contact that doesn't cause hypersensitivity reactions very often. This is different from nickel-containing alloys, which can cause allergic reactions in sensitive groups. This surface chemistry encourages straight bone apposition without fibrous tissue getting in the way. This creates the stable base that is needed for load-bearing implant operation.
Even though cobalt-chromium frameworks work physically, they release ions that can change the color of nearby tissues and cause systemic problems in patients who are vulnerable. Titanium's biological benefits mean fewer complications, lower levels of inflammation markers in the tissues around the device, and better long-term security. These clinical benefits cost makers money because biocompatibility problems lead to device recalls, warranty claims, and damage to their image, which is much worse than any initial material cost savings. When reviewing supplier offers, people in charge of buying things should put a high priority on material certification documents, especially those that show the make-up of the materials and how their surfaces affect biological performance.
Economic Considerations for Volume Procurement
While titanium is more expensive as a raw material than other materials, a total cost study shows more complex economic connections. Compared to harder cobalt-chromium alloys, this material is easier to machine, which cuts down on processing time and tool wear in CAD/CAM production processes. This helps to partly make up for differences in material prices. When makers use less dense materials, they can make more finished goods per kilogram of raw material.
This improves yield economics in situations where a lot of goods are made. Because titanium doesn't rust, it doesn't need a protective covering like stainless steel does. This makes making easier and cuts down on the number of steps needed to complete a process. The material's ability to conduct heat and weld makes production more efficient, especially for unique shapes that need to be changed after they've been machined. The clinical performance benefits may be the most important thing because they translate into market differentiation possibilities.
This means that companies that make titanium-based products can charge higher prices because the results are better. B2B buyers shouldn't just look at how much a unit of material costs; they should also look at the whole package of value that the seller offers, such as how efficiently they handle orders, how consistent the quality is, how well they follow certification rules, and how well they can help with technical issues that affect the overall cost of production.
Procurement Insights: Sourcing High-Quality Implant Titanium Bars
To successfully source materials, you need to know how to navigate the complicated world of medical-grade titanium supply chains, quality systems, and seller qualification methods. Choosing to work with a certain maker has effects that go beyond transaction costs. These effects include following the rules, making sure there is a steady supply of goods, and finally, how well the finished product works.
Critical Supplier Qualification Criteria
Companies that make oral devices need to make sure that the makers of titanium bar dental products keep their certifications up to date so that they meet international medical device standards. ISO 13485:2016 certification shows that thorough quality management systems have been put in place for making medical products. These systems cover everything from being able to track down raw materials to following strict rules for final inspections. ISO 9001:2015 gives basic quality assurance, but it doesn't have the medical-specific rules that are needed for parts that are implanted.
The European CE stamp and the FDA material master file paperwork make sure that the regulations are followed for the target markets. This keeps buyers from having trouble getting into those markets and from being held legally responsible. In addition to certifications, when evaluating a supplier, you should look at their production capabilities, such as their precision machining equipment, metrology systems that can check measurements within certain tolerances, and environmental controls that keep things clean enough for medical materials.
Direct factory checks show things about the company's quality mindset, process discipline, and systems for ongoing growth that certifications alone can't. Buyers can check claims before committing to large-scale relationships by asking for samples of materials along with material certificates, mechanical test results, and surface analysis paperwork.
Transparent Sourcing and Supply Chain Security
Material traceability is a must in medical device supply chains, but many buyers don't look closely enough at the paperwork that shows where the materials came from. For each production lot, premium providers give full mill certificates that list the chemical makeup of the raw materials, the settings for heat treatment, and a check of the mechanical properties. This paperwork helps makers set up lot-level traceability that connects finished products to their source materials.
This is necessary for regulatory filings and monitoring after the product has been sold. Geographic diversity of raw material sources lowers the risk of supply disruptions caused by events in the region, and dual-sourcing methods give you backup plans in case of quality problems or limited capacity. Buyers should look at how suppliers handle their inventory, such as their safety stock policies and lead time promises that help them make regular production schedules.
Long-time suppliers often keep strategic material stocks that protect customers from changes in the market, while younger suppliers may not have the money to make such inventory investments. Look closely at the connection between the prices of raw materials and the prices that are mentioned, because price differences that can't be explained could mean that quality is being compromised or that the business model isn't viable, which creates supply risks in the future.
Customization Capabilities and Technical Partnership
As the need for custom implant options for each patient grows, providers must offer more than just standard materials. OEM services, such as custom diameter tolerances, specialized surface treatments, and pre-machined blank designs, help device makers cut down on the work that needs to be done later and make production more efficient. Suppliers who have their own research and development departments can work together to improve material specifications, create new processes for different shapes, and make quality control methods that are specific to each application.
Technical support that goes beyond transactional relationships, like help with failure analysis, material selection, and regulatory documentation, adds a lot of value and justifies higher prices for buyers who prefer partnership approaches over transactional ones. With more than 20 years of experience making medical titanium products, Baoji INT Medical Titanium Co., Ltd. is a great example of this relationship model because they offer full customization services, from choosing the material grade to making sure the finished dimensions are exactly what you need. Our engineering team works directly with clients to understand their application needs, suggest the best specifications, and offer ongoing technical support during the product development and production scaling stages. This way, we build relationships with our clients that go beyond just supplying materials.
Manufacturing Process and Quality Assurance for Titanium Dental Bars
Knowing how implant titanium bars are made is important for judging the skills of suppliers and guessing how well materials will work. From raw alloy to precision-machined part, the production process has many steps. Each step improves the quality and stability of the end product.
Raw Material Selection and Alloy Preparation
The process of making something starts with carefully choosing titanium sponge and alloying elements that meet medical-grade pure standards. Controlling trace elements is very important because even small impurities can hurt the biocompatibility and functional qualities of a material. Vacuum arc remelting (VAR) or electron beam melting is used on new titanium material to get uniform chemistry and an improved microstructure that doesn't have any macro-segregation.
Multiple remelting processes get rid of flaws and make sure that the makeup is the same across all cross-sections of the ingot. Cast ingots are turned into bar stock with controlled grain structures that make them strong and flexible through hot working processes like casting and rolling. The final mechanical properties are controlled very exactly by the heat treatment steps that change phases and cause precipitation. At every step of the process, parameters must be carefully controlled and checked, and statistical process control systems must keep an eye on key factors to make sure that all production lots are the same. Vertically integrated manufacturers can control these upstream processes better than wholesalers who buy from many mills with different quality systems, so they can make sure that the materials meet stricter standards.
Precision Machining and Surface Treatment
To turn bar stock into final implant frames, you need advanced machining skills that combine multi-axis CNC milling with precise fixturing systems. Computer-aided production software turns prosthesis designs into tool paths that remove materials as quickly and efficiently as possible while keeping the required tolerances for size, which are usually ±0.05 mm. The choice of cutting tool, the supply of coolant, and the best feed rate all help to avoid work hardening and surface damage that could hurt fatigue performance or osseointegration.
After rough grinding, finishing steps are used to get the surface to a certain level of roughness, usually between 0.8 and 1.6 micrometers for areas that will touch implants. Acid passivation and other surface treatment methods get rid of grinding leftovers and improve the titanium oxide layer that helps living things interact with the metal. For some uses, micro-roughening methods that help bones stick together are helpful. These can be done with grit grinding, acid etching, or anodization.
Quality machining centers keep the environment under control so that particulate pollution is kept to a minimum. This is very important because surface cleaning has a direct effect on biocompatibility. Before packing, manufacturers should make sure that cleaning processes are correct by using surface analysis methods to make sure that all organic residues and metal bits have been removed.
Quality Control and Certification Documentation
Full quality control methods tell the difference between high-end providers and vendors of common materials. When raw materials come in, inspection routines check the certificates against the buy specs. Random sampling for independent composition analysis finds changes that haven't been recorded. In-process checks at key points in the manufacturing process stop defects from spreading and allow real-time process changes that keep the product consistent. Final inspection includes checking the sizes using coordinate measure machines or optical comparators, judging the quality of the surface, and going over the paperwork to make sure the lot can be tracked.
Tensile strength, yield strength, and elongation traits are checked mechanically on representative samples to make sure they meet the minimum requirements. The results are written down on material papers that are sent with packages. Corrosion resistance testing mimics the conditions that occur in living things, proving that the inactive film is stable and there is no limited attack. Biocompatibility validation using ISO 10993 biological evaluation methods gives proof that supports regulatory applications and customer quality needs.
The best makers keep these huge quality records in digital systems that make them easy to get back for customer audits, regulatory questions, or reviews after the product has been sold. Baoji INT Medical Titanium Co., Ltd. has quality systems that are approved to both ISO 9001:2015 and ISO 13485:2016 standards. Their products also have the EU CE stamp, which means they follow the rules for medical devices in Europe. Our quality paperwork packages show our clients that we are committed to being open and honest, from the source of the raw materials to the final inspection. This helps them with their own regulatory reports and quality system standards.
Conclusion
Implant titanium bars are a revolutionary technology in prosthodontics that overcomes the main problems with traditional dentures by providing better physical stability, load distribution, and osseointegration. Material benefits of medical-grade titanium metals, such as excellent biocompatibility, the best strength-to-weight ratios, and amazing corrosion resistance, make titanium the best choice for making implant bars. When looking for these important parts, procurement workers should give more weight to sellers who can show they have all the necessary quality certifications, material traceability systems, and expert support skills. When you know about the manufacturing processes, material requirements, and clinical performance drivers, you can make smart sourcing choices that balance the original costs with the long-term value that comes from better results and fewer complications.
FAQ
Why are titanium bars better at keeping dentures in place than other holding systems?
Titanium bar frames make rigid links between several implants. This spreads the biting forces evenly across all fixtures instead of putting most of the load on a few places. This load-sharing system lowers stress at the point where the bone meets the implant and almost completely stops the tiny movements that hurt tissues and cause bone loss in regular dentures. The high elastic stiffness and yield strength of the material keep its shape even when it is loaded and unloaded many times. This means that the material will continue to fit perfectly for years to come.
Can titanium dental bars be customized for different implant configurations?
Modern CAD/CAM production makes it possible to completely change the shape of titanium bars to fit the anatomy, implant sites, and prosthetic designs of each patient. Bars can be made with different cross-sectional shapes, connection housing places, and screw access arrangements to fit different medical needs. This adaptability allows for a wide range of dental restorations, from simple four-implant mandibular overdentures to full-arch rehabilitations with curved implants and custom emerging profiles.
How can buyers make sure that certificates and quality standards are met?
Reliable sellers offer complete paperwork packages that include mill certificates that confirm the alloy's composition, mechanical test results that confirm its strength qualities, and biocompatibility certifications that show ISO 10993 compliance. Ask for material papers that are specific to the lots that are being bought, not general specification sheets. Check that the supplier has the right quality system certifications, like ISO 13485:2016 for medical products, and that they have the right legal clearances, like CE marking or FDA registration, for the markets they want to reach.
Partner with a Trusted Titanium Bar Dental Manufacturer
To improve your dental implant product line, you need to work with a titanium bar dental provider that is dedicated to quality, innovation, and customer satisfaction. Baoji INT Medical Titanium Co., Ltd. has been making medical-grade titanium products for more than 20 years and makes high-quality Grade 5 and Grade 23 metal bars that meet the strictest performance and licensing standards. We can make custom diameters up to 200 mm, lengths up to 3000 mm, and OEM services that are made to fit the needs of your individual purpose.
Each item has full ISO 9001:2015, ISO 13485:2016, and EU CE certifications, along with full tracking documents and technical advice services. Our team is ready to help you reach your goals with consistent quality, on-time delivery, and cheap pricing, whether you're making new implant systems, increasing production volumes, or looking for a trusted supply chain partner. Send us an email at export@tiint.com to talk about your needs and find out why Baoji INT Medical Titanium Co., Ltd. is the titanium bar dental supplier of choice for top dental device makers around the world.
References
1. Misch, C.E. (2015). Dental Implant Prosthetics, 2nd Edition. Elsevier Health Sciences.
2. Brunski, J.B. (2018). "Biomechanical Considerations in Dental Implant Design and Clinical Performance." International Journal of Oral & Maxillofacial Implants, 33(4): 127-145.
3. Niinomi, M., Nakai, M., and Hieda, J. (2012). "Development of New Metallic Alloys for Biomedical Applications." Acta Biomaterialia, 8(11): 3888-3903.
4. Thomason, J.M. (2012). "The Use of Mandibular Implant-Retained Overdentures Improve Patient Satisfaction and Quality of Life." Journal of Dental Research, 91(7): 591-597.
5. Att, W., Bernhart, J., and Strub, J.R. (2009). "Fixed Rehabilitation of the Edentulous Maxilla: Possibilities and Clinical Outcome." Journal of Oral and Maxillofacial Surgery, 67(11): 60-73.
6. International Organization for Standardization (2016). ISO 5832-3: Implants for Surgery — Metallic Materials — Part 3: Wrought Titanium 6-Aluminium 4-Vanadium Alloy. Geneva: ISO Standards.