What are the uses of titanium bars in the medical field?
 2026-04-21 09:09:12
2026-04-21 09:09:12
Medical-grade titanium bars are an important part of making surgical implants and other high-tech medical tools. The titanium bar for medical use, especially Grade 5 (Ti-6Al-4V) and Grade 23 (Ti-6Al-4V ELI), is very strong, doesn't rust, and is biocompatible. Vacuum arc remelting is used to make sure that the microstructures are intact and that there are few impurities. Because of this, these special bars are needed for surgery tools, spine fusion devices, orthopedic implants, and oral prosthetics. Their special mix of being light, strong, and biologically safe solves important problems in fixed implantation and long-term patient results.
|
|
|
Understanding Titanium Bars for Medical Use
What Defines Medical-Grade Titanium Bars
Medical-grade titanium bars are metal pieces that are carefully designed and made to strict standards for use in healthcare. Tough quality rules and chemical limits make these materials very different from industrial-grade titanium. Grade 23 titanium, also known as "Extra Low Interstitial" titanium, has oxygen levels below 0.13% and iron levels below 0.25%. This makes it more flexible and harder to break than normal Grade 5 alloys.
During the manufacturing process, vacuum arc remelting methods get rid of high-density spots and make sure that the microstructure of the bar is the same all the way through its cross-section. When making complicated shapes for implanted devices, this uniformity is very important because any difference in the material could weaken the structure or make the device less effective in the body.
Key Alloy Compositions and Material Standards
There are two main types of titanium alloys used in the medical field: pure titanium (Grades 1-4) and Ti-6Al-4V forms (Grades 5 and Grade 23). Pure titanium types are biocompatible to the highest degree, but they are also weaker mechanically, making them good for uses that need very little load-bearing capacity. Ti-6Al-4V alloys have a tensile strength of more than 860 MPa and a low elastic stiffness (~110 GPa), which is similar to how bones work.
When buying medical supplies, following foreign norms is still a must. The chemical make-up, mechanical qualities, and tracking rules are set by ASTM F136 and ISO 5832-3. These licenses make sure that every batch meets strict quality standards, which are checked by mill test certificates that meet EN 10204 3.1 documentation standards. Purchasing managers give more weight to suppliers who consistently follow these rules, since gaps in certification can stop whole production processes during checks.
Medical Applications of Titanium Bars
Orthopedic Implants and Trauma Fixation
The main area where medical titanium bars are used is in orthopedic treatment. These materials can be changed into bone plates, intramedullary nails, and fracture fixing screws that help bones work again after they've been hurt. The high strength-to-weight ratio keeps the implant's weight down while still giving it enough power to support bone structures as they heal. Titanium is preferred by surgeons over stainless steel because its elastic stiffness is very close to that of real bone.
This compatibility lowers stress shielding phenomena, which happens when implants that are too stiff stop the bone from remodeling properly. This leads to better long-term integration. Spinal rods, hip replacement stems, and knee joint parts are all made from titanium bars that are carefully cut and then cold-worked for a long time to increase their yield strength without losing their flexibility.
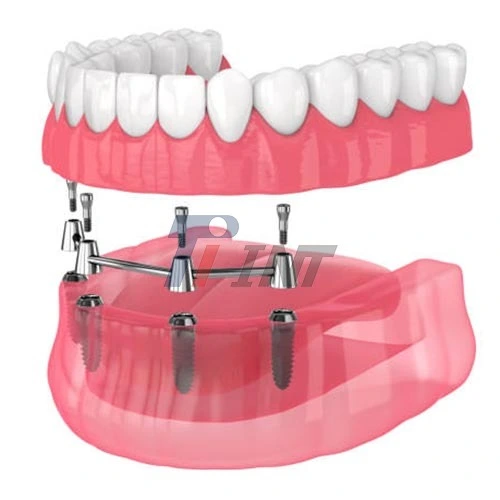
Dental Implantology and Craniofacial Reconstruction
For dental uses, titanium bars need to have a very smooth surface and be exactly the right size. Implant abutments and dental screws made from these bars need to have threads that are accurate to the micron level so that bacteria don't get into the space between the bone and the implant. The mouth is a very tough place to work on teeth because the pH levels change all the time and there are enzymes that break down less durable materials.
In addition to regular tooth implants, craniofacial surgery uses custom-made metal parts for people who need a lot of work done on their faces. Titanium mesh and plates are shaped by surgeons to fix problems with the orbital floors, the mandible, and the head. The non-magnetic qualities of the material make it compatible with MRI, which means that imaging after surgery can be done without artifacts or patient pain, especially when considering titanium bar for medical use.
Surgical Instruments and Specialized Tools
Titanium bars are machined by companies that make medical instruments into high-precision surgery tools like knife handles, retractors, and special forceps. Titanium is light, which makes these tools useful because it keeps surgeons from getting tired during long treatments. The material doesn't rust and can be sterilized in a sterilizer many times without breaking down. This makes tools last longer and saves healthcare facilities money on replacement costs.Swiss-style CNC machining centers can work with titanium bars that have ISO h7 or h8 specs.
This makes it possible to make the complex shapes needed for minimally invasive surgery devices. For these machines' guide rings to stay stable during high-speed cutting, the diameters must be very close to each other. This level of accuracy in production directly turns into the dependability of the instruments used in important surgeries.
Benefits of Using Titanium Bars in the Medical Industry
Superior Biocompatibility and Osseointegration
Titanium is biocompatible because it has a stable oxide layer that grows on its own when it is introduced to air. This passive film stops the release of metallic ions into the nearby tissue. This eliminates the risk of allergic reactions and rejection that come with metals that contain cobalt, chromium, or nickel. Osseointegration rates for titanium implants are higher than 95%, which means that bone tissue grows straight onto the implant surface without being surrounded by fibrous tissue.
The fact that the material doesn't mix with anything makes it especially useful for people who are sensitive to metals or have allergic diseases. Unlike stainless steel, which contains nickel, titanium alloys cause very little inflammation. This speeds up the healing process and lowers the risk of problems after surgery. Because it is biologically compatible, titanium is usually used for permanent implants that will stay in the body for decades.
Exceptional Corrosion Resistance and Longevity
Body fluids produce an environment that is acidic and full of chloride, which constantly tests implant materials. Titanium's resistance to pitting corrosion and crevice corrosion makes sure that the structure stays strong for long amounts of time after insertion. Titanium's mechanical qualities stay the same after decades of imagined physiological exposure, as shown by tests done in the lab.
This longevity means that healthcare systems can save money. Revision treatments are very expensive and come with a lot of risks for the patient when implants fail. Titanium's durability lowers the number of revisions needed, which lowers the total cost of treatment while raising the quality of life for patients. The stability of the material also makes it easier for device makers to keep track of their supplies, since products stay fresh without any special storing needs.
Optimal Strength-to-Weight Ratio and Fatigue Resistance
Titanium bars have a final tensile strength of about 1000 MPa when they are cold-worked, and they weigh about 40% less than stainless steel parts that do the same job. This balance between strength and weight lets implant makers cut down on the weight of parts without affecting their structural stability. When implants are lighter, they don't irritate soft tissues as much and make daily tasks more comfortable for the patient.For load-bearing devices that are put through millions of cycle stress events, fatigue resistance is very important.
Every year, a hip replacement goes through about one million walking rounds, which is a lot of stress that wears down weak materials over time. Titanium has a high-cycle fatigue strength, which means that implants can handle these stresses without cracks spreading or breaking suddenly. Before a device is approved by the government, it is tested for fatigue performance using accelerated lifetime models.
How to Choose the Right Titanium Bar for Medical Use
Selecting Appropriate Alloy Grades
The first step in making a purchase choice is to fit the titanium grade to the needs of the application. Grade 23 (Ti-6Al-4V ELI) is the best material for implants that are likely to break and cause terrible results if they do fail. Its increased flexibility and toughness give it extra protection against loads that aren't expected or mistakes in the making process. Grade 5 titanium is about 15% to 20% cheaper, but it has more intermediate elements, which make it less flexible. Grades 2–4 of pure titanium are best for uses that need the most biocompatibility with low mechanical needs.
These types are often used in dental abutments and maxillofacial plates because they are more corrosion-resistant and easier to shape than they are strong. To make sure the material is right for the job, R&D engineers should ask for material property data sheets that list the tensile strength, yield strength, stretch percentage, and elastic modulus, especially when considering titanium bar for medical use.
Evaluating Manufacturing Capabilities and Certifications
Dependability in the supply chain is what sets good sellers apart from great partners. Medical device companies should check the quality control systems of possible suppliers and make sure they are ISO 13485:2016 certified. They should also check the suppliers' production capacities. Questions about vacuum arc remelting, heat treatment routines, and surface finishing methods show how advanced the production process is.Suppliers who offer complete tracking paperwork have an edge when it comes to regulatory reports.
Full mill test records that show the material's journey from the ingot to the finished bar size meet FDA 21 CFR Part 820 standards and EU Medical Device Regulation 2017/745 requirements. When evaluating vendors, purchasing managers should ask to see sample certificates to make sure that all the necessary paperwork is included before signing supply deals.
Customization Options and Small-Batch Feasibility
More and more, new medical technologies need specialized parts that are only available in small amounts. Suppliers who can make unique sizes, lengths, and standards without having very high minimum order quantities make product development more flexible. Baoji INT Medical Titanium Co., Ltd. can handle small-batch production for new medical device businesses, helping with research and development (R&D) through pilot production without making customers commit to too much inventory.
Customizing the surface process adds more value. Anodizing makes controlled oxide layers that improve the qualities of osseointegration and allow color-coding to show the size of the part. Centerless grinding can get surface finishes close to 0.4 Ra, so Swiss-machined parts don't need any extra steps of processing. These customization options cut down on the time it takes for new gadgets to hit the market while keeping costs low.
Trends and Innovations in Medical Grade Titanium Bars
Advanced Manufacturing Technologies
For certain uses, additive manufacturing methods can now be used along with standard bar stock machining. By printing titanium alloys in three dimensions, it is possible to make implant shapes that are specific to each patient, which is not possible with traditional subtractive methods. Powder bed fusion processes create porous structures that encourage bone growth. This makes implants more stable when bone quality is low.Precision CNC cutting keeps getting better with the addition of multiple axes and real-time process tracking.
Modern machining centers with adaptable control systems change the cutting settings on the fly to account for changes in the material and make the tools last longer. These technology advances lower the cost per unit while improving the accuracy of measurements. This makes it possible for more patients to afford more complicated implant designs.
Enhanced Surface Treatments and Bioactive Coatings
Surface modification technologies change the way implants and tissue connect, which speeds up the process of osseointegration. Acid etching makes micro-rough surfaces that help proteins stick to surfaces and cells stick to them more easily. Plasma spray coatings put down hydroxyapatite layers that look like natural bone minerals. However, titanium is naturally biocompatible, so coatings aren't usually needed for most uses.Nanostructured surface treatments are the next big thing.
Titanium dioxide nanotube arrangements that are biocompatible and have antibacterial qualities are made through controlled oxidation processes. These new ideas reduce the chance of getting an illness after surgery without using antibiotic-eluting coatings that lose their usefulness over time. These developments are moving closer to clinical use thanks to research institutions working with material sources.
Global Supply Chain Evolution
Geographical trends of output show how competitive benefits in processing titanium change over time. Asian makers, especially Chinese suppliers with decades of experience in the titanium business, offer options that are both cheaper and better in terms of quality. Companies like Baoji INT Medical Titanium Co., Ltd., which was formed in 2003 and has more than 30 years of experience in the field, show how quality investments over time can help regional providers become competitive on the international market.
Buyers in North America and Europe are becoming more aware of worth that goes beyond unit price. Supply chain stability, expert help, and the quality of regulatory documents often matter more than small differences in cost. Successful providers offer application engineering help, which helps customers choose the best materials and set the best processing settings. This method of consulting builds long-lasting relationships that can withstand changes in the market and in the rules that govern them, particularly when it comes to titanium bar for medical use.
Conclusion
Medical-grade titanium bars are an essential part of modern healthcare manufacturing because they make it possible for life-changing implants and surgery innovations. Their special mix of biocompatibility, mechanical strength, and resistance to rust solves some of the most important problems in permanent implants. Competitive pricing isn't the only thing that procurement professionals should look for in providers; they should also look for thorough certifications, manufacturing skills, and a technical support infrastructure. The medical titanium business keeps growing thanks to new ways of making the metal and treating its surface, which allow for more uses. For device development to go smoothly, it's important to work with material providers who have a lot of knowledge, know the rules, and can guarantee consistent quality through strict process controls.
FAQ
Why choose Grade 23 over Grade 5 titanium for implants?
Grade 23 titanium has tightly managed interstitial elements, with oxygen levels below 0.13% and iron levels below 0.25%. This makes it more flexible and harder to break than normal Grade 5 alloys. This better performance is very important for fracture-critical implants, where a loss of the material could do a lot of damage to the patient. The extra low interstitial makeup makes it about 15-20% more expensive to make, but medical device rules usually require Grade 23 for permanent implants that hold a lot of weight.
What certifications must accompany medical titanium bar shipments?
Minimum paperwork needs to include mill test papers that meet EN 10204 3.1 standards. These certificates need to list the chemicals used, their mechanical qualities, the conditions of the heat treatment, and a statement that they meet the requirements of ASTM F136 or ISO 5832-3. Biocompatibility testing reports and production process validation records may be needed as extra paperwork, based on the final use and the rules of the market where the product will be sold.
Can suppliers accommodate small customized orders for prototyping?
Medical titanium providers with a good reputation are supporting small-batch production more and more to make room for new ideas. When sellers keep their manufacturing options open, they can make things with custom diameters, lengths, and tolerances without having to meet huge minimum order amounts. This method lets medical device startups and research centers make new goods without having to spend a lot of money on inventory during the creation stages.
Partner with Baoji INT Medical Titanium Co., Ltd. for Your Medical Device Success
If a medical device maker needs an approved, high-performance titanium bar for medical use, they can get better results by working with experts in the field. Baoji INT Medical Titanium Co., Ltd. has been handling titanium for more than 30 years and has been working in the medical field since 2003. They have full ISO 9001:2015, ISO 13485:2016, and EU CE certifications. Our full line of products includes pure titanium rods, Ti-6Al-4V ELI bars, and custom-forged parts made to fit the needs of orthopedic, dental, and medical instruments.
We know the problems that R&D engineers and supply chain managers face when they have to buy things, like having to meet strict safety standards and tight delivery deadlines. Our expert team can help you choose the right materials, give you advice on how to make things that can be machined, and give you full traceability paperwork that makes regulatory submissions easier. Our manufacturing options are open enough to fit projects at any stage of development, whether they need help with small-batch prototypes or large-scale production runs.Email our team at export@tiint.com to talk about your particular needs. We show our dedication to quality and partnership by giving you thorough technical specs, cheap quotes, and samples of our materials.
References
1. Rack, H.J., & Qazi, J.I. (2012). Titanium alloys for biomedical applications. Materials Science and Engineering C, examining mechanical properties and biocompatibility of medical-grade titanium alloys.
2. Geetha, M., Singh, A.K., Asokamani, R., & Gogia, A.K. (2009). Ti based biomaterials, the ultimate choice for orthopedic implants – A review. Progress in Materials Science, comprehensive analysis of titanium applications in medical devices.
3. Long, M., & Rack, H.J. (1998). Titanium alloys in total joint replacement—a materials science perspective. Biomaterials, investigating clinical performance of titanium implants.
4. Niinomi, M. (2008). Mechanical biocompatibilities of titanium alloys for biomedical applications. Journal of the Mechanical Behavior of Biomedical Materials, evaluating stress shielding and osseointegration factors.
5. Liu, X., Chu, P.K., & Ding, C. (2004). Surface modification of titanium, titanium alloys, and related materials for biomedical applications. Materials Science and Engineering R, covering surface treatment technologies for enhanced implant performance.
6. Elias, C.N., Lima, J.H., Valiev, R., & Meyers, M.A. (2008). Biomedical applications of titanium and its alloys. JOM Journal of the Minerals, Metals and Materials Society, discussing manufacturing processes and clinical applications of medical titanium products.